Page 90 - Innovator's Science-8
P. 90
Melting points of some solids
Science S.No. Material Mel ng point
Bytes
Testimony
Every substance melts at a different
1. Ice 0°C
temperature, known as its mel ng point. Ice
melts at 0°C, urea at 133°C, and iron at a very
high temperature of 1538°C. Water, like all
liquids, does not have a fixed shape - it takes the
shape of the container into which it is poured.
This is because the par cles of a liquid can move 2. Urea 133°C
freely and slide past each other. However, liquids
have a definite volume, which means that even if
the shape of the container changes, the quan ty
of water remains the same. The par cles in a
liquid are close enough to stay together but can
3. Iron 1538°C
s ll move around, giving the liquid a fixed
volume but no fixed shape.
Activity-1
Experiential Learning
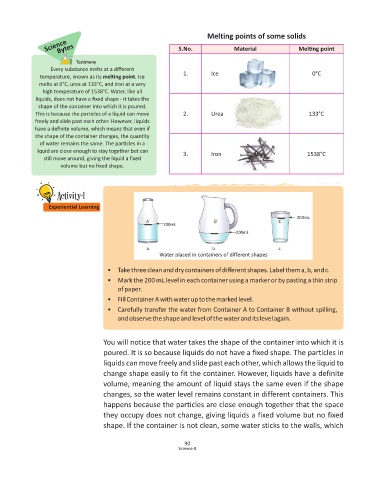
200mL
200mL
200mL
a. b. c.
Water placed in containers of different shapes
• Take three clean and dry containers of different shapes. Label them a, b, and c.
• Mark the 200 mL level in each container using a marker or by pas ng a thin strip
of paper.
• Fill Container A with water up to the marked level.
• Carefully transfer the water from Container A to Container B without spilling,
and observe the shape and level of the water and its level again.
You will notice that water takes the shape of the container into which it is
poured. It is so because liquids do not have a fixed shape. The particles in
liquids can move freely and slide past each other, which allows the liquid to
change shape easily to fit the container. However, liquids have a definite
volume, meaning the amount of liquid stays the same even if the shape
changes, so the water level remains constant in different containers. This
happens because the particles are close enough together that the space
they occupy does not change, giving liquids a fixed volume but no fixed
shape. If the container is not clean, some water sticks to the walls, which
90
Science-8