Page 92 - Innovator's Science-8
P. 92
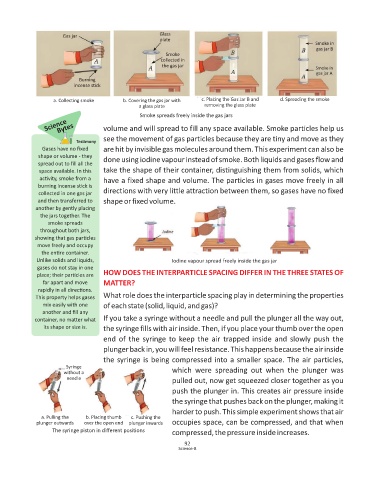
Gas jar Glass
plate
Smoke in
gas jar B
Smoke
collected in
the gas jar
Smoke in
gas jar A
Burning
incense stick
a. Collecting smoke b. Covering the gas jar with c. Placing the Gas Jar B and d. Spreading the smoke
a glass plate removing the glass plate
Smoke spreads freely inside the gas jars
Science volume and will spread to fill any space available. Smoke particles help us
Bytes
see the movement of gas particles because they are tiny and move as they
Testimony
Gases have no fixed are hit by invisible gas molecules around them. This experiment can also be
shape or volume - they done using iodine vapour instead of smoke. Both liquids and gases flow and
spread out to fill all the
space available. In this take the shape of their container, distinguishing them from solids, which
ac vity, smoke from a have a fixed shape and volume. The particles in gases move freely in all
burning incense s ck is
collected in one gas jar directions with very little attraction between them, so gases have no fixed
and then transferred to shape or fixed volume.
another by gently placing
the jars together. The
smoke spreads
throughout both jars,
showing that gas par cles
move freely and occupy
the en re container.
Unlike solids and liquids, Iodine vapour spread freely inside the gas jar
gases do not stay in one
place; their par cles are HOW DOES THE INTERPARTICLE SPACING DIFFER IN THE THREE STATES OF
far apart and move MATTER?
rapidly in all direc ons.
This property helps gases What role does the interparticle spacing play in determining the properties
mix easily with one of each state (solid, liquid, and gas)?
another and fill any
container, no ma er what If you take a syringe without a needle and pull the plunger all the way out,
its shape or size is. the syringe fills with air inside. Then, if you place your thumb over the open
end of the syringe to keep the air trapped inside and slowly push the
plunger back in, you will feel resistance. This happens because the air inside
the syringe is being compressed into a smaller space. The air particles,
Syringe
without a which were spreading out when the plunger was
needle
pulled out, now get squeezed closer together as you
push the plunger in. This creates air pressure inside
the syringe that pushes back on the plunger, making it
harder to push. This simple experiment shows that air
a. Pulling the b. Placing thumb c. Pushing the
plunger outwards over the open end plunger inwards occupies space, can be compressed, and that when
The syringe piston in different positions compressed, the pressure inside increases.
92
Science-8